|
coli HPAH (HpaC) is a flavin reductase generating free reduced FAD that is transferred to the larger component (HpaB) to hydroxylate HPA (3, We have isolated HPAH from Acinetobacter baumannii and showed that the enzyme is quite different from the analogous HPAH enzymes from both P. A different HPAH system was later isolated from Escherichia coli W, and studies have shown that the smaller component of E. putida HPAH seems to be similar to the mechanism of single-component aromatic flavoprotein hydroxylases except that two proteins are required (5-7). The reaction with NADH and that with oxygen and substrate were reported to occur on the flavoprotein component. putida HPAH have shown that FAD is tightly bound to the smaller component and that the larger component appears to be a coupling protein enabling hydroxylation (2, 5). 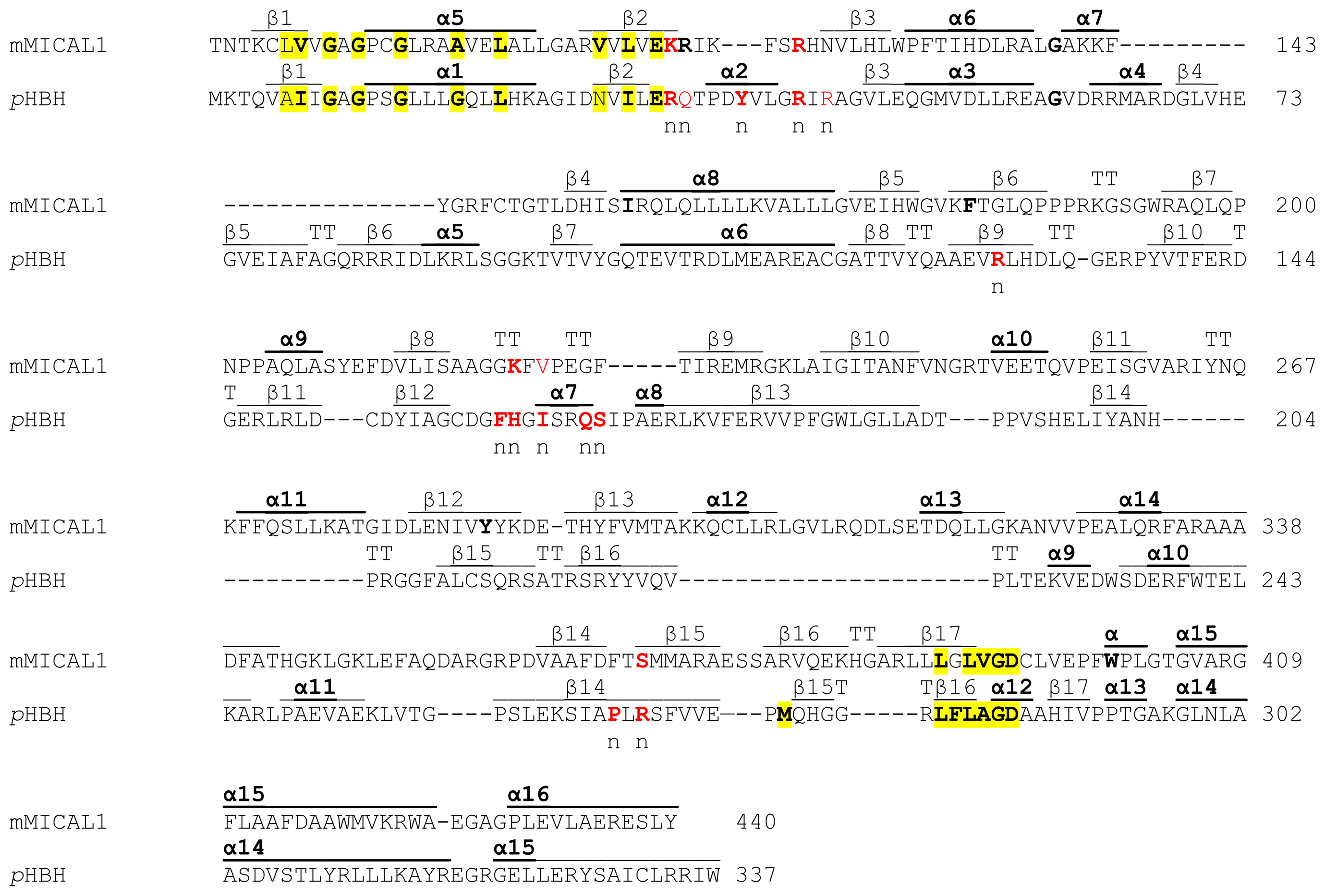
The first HPAH purified was from Pseudomonas putida and shown to be a two-protein component enzyme system (2). In the past decade, it has been revealed that the hydroxylation of HPA in various organisms is carried out by at least three different types of two-protein component enzyme systems (2-4). This hydroxylation is usually found as the initial step in the aerobic degradation pathway for HPA, one of the major metabolites of lignin (1). p-Hydroxyphenylacetate hydroxylase (HPAH) 1 catalyzes hydroxylation of p-hydroxyphenylacetate (HPA) to form 3,4-dihydroxyphenylacetate (DHPA). Rate constants for both phases were the same in the absence and presence of HPA, but in the presence of HPA, the equilibrium shifted toward the faster reacting species. In the reaction of reduced C 1 with oxygen, the reoxidation reaction is also biphasic, consistent with reduced C 1 being a mixture of fast and slow reacting species. 
When the C 1 -HPA complex was reduced by 4(S)-NADH, the reduction rate was changed from 300 to 30 s -1, giving a primary isotope effect of 10 and indicating that C 1 is specifically reduced by the pro-(S)-hydride. The redox potentials indicate that the rate enhancement is not due to thermodynamics (E°m of the C 1 -HPA complex is -245 mV compared to an E°m of C 1 of -236 mV). Formation of the C 1 -HPA complex prior to binding of NADH was required for this stimulation. However, in the presence of HPA, the reduction of C 1 by NADH occurred in a single phase at 300 s -1 with a K d of 25 µM for NADH binding at 4☌. This result indicates that C 1 exists as a mixture of isoforms. Reduction of C 1 by NADH occurs in two phases with rate constants of 11.6 and 3.1 s -1 and K d values for NADH binding of 2.1 and 1.5 mM, respectively. The apoenzyme of C 1 binds to oxidized FMN tightly with a K d of 0.006 µM at 4☌, but with a K d of 0.038 µM in the presence of HPA. We have shown here that HPA plays important roles in the reaction of C 1. 
C 1 is a flavoprotein containing FMN, and its function is to provide reduced flavin for C 2 to hydroxylate HPA. It is a two-protein system consisting of a smaller reductase component (C 1 ) and a larger oxygenase component (C 2 ). ABSTRACT: p-Hydroxyphenylacetate (HPA) hydroxylase (HPAH) from Acinetobacter baumannii catalyzes hydroxylation of HPA to form 3,4-dihydroxyphenylacetate.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
